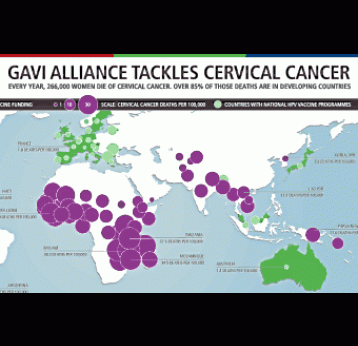
Gavi’s impact
Since the inception of Gavi’s human papillomavirus (HPV) vaccine programme in 2012, 3.9 million girls have been protected against the leading cause of cervical cancer, and demand for HPV vaccine has never been higher
Record Demand
In 2016, the Gavi Board approved an acceleration of the HPV vaccine programme, allowing countries in the first year of their programme to scale up directly and vaccinate multi-age cohorts of girls in the age range 9–14 years. Through this new strategy, Gavi aimed to protect around 40 million girls from cervical cancer by 2020, averting an estimated 900,000 deaths. However, due to surging global demand and vaccine supply shortages, the scope of this goal has been reduced to reaching an estimated 14 million girls and averting 300,000 deaths.
The uptake of the HPV programme since inception has been encouraging, with 27 countries approved for Gavi support – of which 18 have successfully introduced HPV vaccine into their national immunisation programme. These countries include those with the highest burden of cervical cancer in the world such as Malawi, Uganda, the United Republic of Tanzania, Zambia and Zimbabwe.
Record low price
In 2011, a record low price for HPV vaccines created an opportunity for low-income countries to vaccinate millions of girls against the leading cause of cervical cancer. Thanks to the Vaccine Alliance, the lowest-income countries now have access to HPV vaccines for as little as US$ 4.50 per dose. The same vaccines can cost more than US$ 100 in high-income countries, and the previous lowest public sector price was US$ 13 per dose. In addition, the World Health Organization (WHO) decision to switch from a recommended schedule of three doses to two doses helps to facilitate country roll-outs and reduce costs.

Gavi’s response
Historically, the high cost of the vaccine and challenges of reaching adolescent girls to deliver immunisation have been barriers to introduction in low-income countries. Gavi is working to bridge the equity gap by providing support for HPV vaccine and ensuring sustainable prices.
Gavi supports HPV vaccines for national introduction, with immunisation of multi-age cohorts of girls in the age range 9–14 years. However, due to ongoing supply constraints, countries have had to adapt their programmatic strategy by vaccinating a single cohort to introduce and the remaining multi-age cohort when supply is available.
WHO recommends that countries use HPV delivery strategies that are compatible with their health systems infrastructure and cold-chain capacity; are affordable, cost-effective and sustainable; and achieve the highest possible coverage. The highest priority should be given to vaccination strategies that include populations who are less likely to have access to screening for cervical cancer later in life. Opportunities to link vaccine delivery to other health interventions targeting adolescent girls should also be explored.
Support
In 2016, the Gavi Board approved two main changes in the HPV programme, following a recommendation by the WHO Strategic Advisory Group of Experts (SAGE) on Immunization.
First, countries can now apply directly for Gavi support to fund national introductions rather than starting with a demonstration programme. Countries also have the option of a phased introduction.
Second, countries can opt to vaccinate multiple age groups – between 9 and 14 years – in the first year of their programme, depending on supply availability.
Partnerships
Gavi works with cancer groups, as well as reproductive health and women’s organisations, to help countries deliver HPV vaccines cost-effectively. These partnerships also identify opportunities to integrate HPV vaccination with other health interventions for girls. These include adolescent reproductive health, HIV prevention, nutrition, family planning and safe motherhood. Where feasible, they have also helped to push through comprehensive cervical cancer prevention plans which include vaccination of young girls and screening and treatment of women.
Partners
Many organisations are actively involved with clinical and operational research, policy analysis and advocacy related to HPV vaccine. Gavi’s collaborating partners include but are not limited to:
- Centers for Disease Control and Prevention (CDC)
- Clinton Health Access Initiative (CHAI)
- Girl Effect
- International Agency for Research on Cancer (IARC)
- Jhpiego
- JSI
- PATH
- SEWA
- UNICEF
- Union for International Cancer Control (UICC)
- United Nations Population Fund (UNFPA)
- vaccine manufacturers and academia
- WHO
The issue
Globally, one woman dies of cervical cancer every two minutes
Cervical cancer is the leading cause of cancer death among women in Gavi-supported countries.

Globally, one woman dies of cervical cancer every two minutes
Cervical cancer is the leading cause of cancer death among women in Gavi-supported countries.
The growing burden of cervical cancer
Infection with human papillomavirus (HPV) is the main cause of cervical cancer, which claims the lives of 311,000 women each year, mainly in low-income countries.
Without changes in prevention and control, cervical cancer deaths are forecast to rise to 416,000 by 2035.
Immunisation against HPV infection, coupled with screening and treatment, is the best strategy to reduce rapidly the burden of cervical cancer. In low-income countries where women often lack access to cancer screening and treatment services, immunising girls before exposure to HPV is critical.
Safe and effective HPV vaccines can prevent up to 90% of all cervical cancer cases.
Surging demand
The year 2018 saw a significant supply shortage of HPV vaccine, which is forecast to continue in the short- to medium-term. This is because of the unprecedented uptake of HPV support in Gavi-eligible countries, combined with increased global demand for HPV vaccines from higher-income countries.
Alliance partners have been working with manufacturers in order to increase manufacturing capacity to meet worldwide demand, as well as working with countries to help them adapt programmatic strategies.
External links
Resources
How to apply for vaccine support
Eligible countries can access detailed guidelines to manage the support they receive from Gavi.